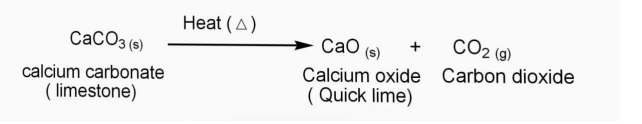 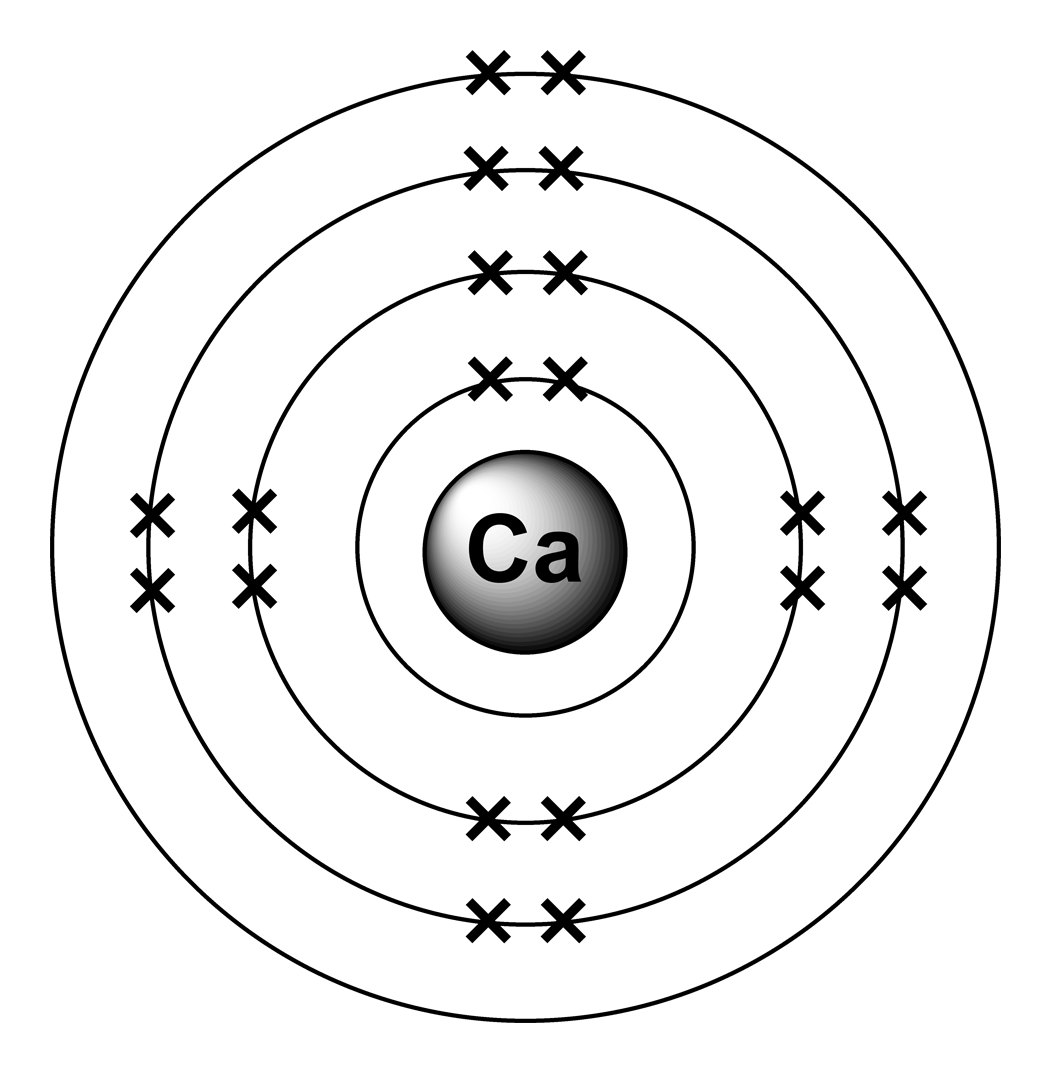
This makes calcium a positive ion with a charge of 2+. … Since calcium lost two electrons, it has 20 protons, but only 18 electrons. What will be the charge of calcium when it becomes an ion?Ĭalcium loses 2 electrons when it becomes an ion. Why is the calcium ion (Ca2+) more stable than the calcium atom (Ca)? The calcium atom has 2 electrons in a valence level above a stable noble gas configuration-If the atom can change into an ion identical to that of a noble gas, it can achieve the stability of the noble gas. Why is a calcium ion more stable than a calcium atom? 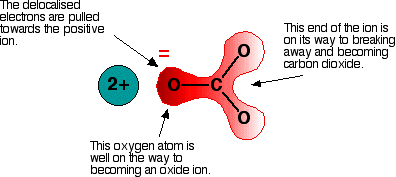
We can conclude that the number of electrons the metal gives and non metal takes is referred to as its charge. Therefore it gives 2 electrons to other non-metal which has 6 electrons in its outermost shell. Read More: What are the four main processes of social cognition? Why does the calcium ion have a charge of +2?Ĭalcium has 2+ charge as it’s electronic configuration is 2,8,8,2. How are calcium ions transported into the cell?Ĭells use calcium pumps to direct the flow of calcium ions through the plasma membrane or organelle membranes, and the resulting gradients are used in a variety of signalling systems mediated by gated ion channels. They make their entrance into the cytoplasm either from outside the cell through the cell membrane via calcium channels (such as calcium-binding proteins or voltage-gated calcium channels), or from some internal calcium storages such as the endoplasmic reticulum and mitochondria. A calcium atom has 20 protons and 20 electrons. The completed passage is: Ca 2 + represents an ion with 20 protons and 18 electrons. If an atom loses electrons, the ion formed in positively charged and is called a cation. When a calcium atom forms an ion It is known as the ion? This results in a cation with 20 protons, 18 electrons, and a 2+ charge. How does calcium become a monatomic cation?įor example, a neutral calcium atom, with 20 protons and 20 electrons, readily loses two electrons. … Positive and Negative Ions: Cations and Anions. The VA elements gain three electrons to form anions with a 3- charge. The VIA elements gain two electrons to form anions with a 2- charge. Does calcium become a cation or anion?Īnd all of them form an anion with a single negative charge. How does calcium become a stable ion?Ĭalcium will lose two valence electrons to achieve the stable configuration of argon. … This simply means that the element calcium needs two more electrons to equal the number of protons that it has this balances the charge, or makes it zero. An ion is an atom of a chemical element that has an unequal number of electrons compared to protons. Why is calcium an ion?Ĭalcium also has a positive charge of 2. When these electrons are lost, a calcium ion, Ca 2 +, is formed.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
